NaHSO4和Ba(OH)2反应Ba(OH)2过量:NaHSO4+Ba(OH)2=BaSO4↓+H2O+NaOH H+ + SO42- + Ba2+ + OH- = BaSO4↓+H2O 按反应来看,最后产物中有NaOH,而Ba(OH)2的碱性要比NaOH的碱性要更弱,不是强碱制弱碱吗?为什么这里是这
来源:学生作业帮助网 编辑:作业帮 时间:2024/07/12 22:11:31
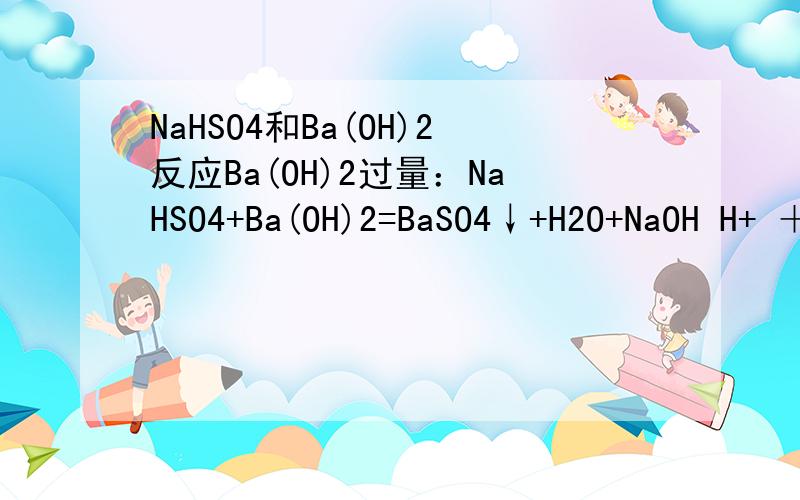
NaHSO4和Ba(OH)2反应Ba(OH)2过量:NaHSO4+Ba(OH)2=BaSO4↓+H2O+NaOH H+ + SO42- + Ba2+ + OH- = BaSO4↓+H2O 按反应来看,最后产物中有NaOH,而Ba(OH)2的碱性要比NaOH的碱性要更弱,不是强碱制弱碱吗?为什么这里是这
NaHSO4和Ba(OH)2反应
Ba(OH)2过量:
NaHSO4+Ba(OH)2=BaSO4↓+H2O+NaOH
H+ + SO42- + Ba2+ + OH- = BaSO4↓+H2O
按反应来看,最后产物中有NaOH,而Ba(OH)2的碱性要比NaOH的碱性要更弱,不是强碱制弱碱吗?为什么这里是这样呢?
NaHSO4和Ba(OH)2反应Ba(OH)2过量:NaHSO4+Ba(OH)2=BaSO4↓+H2O+NaOH H+ + SO42- + Ba2+ + OH- = BaSO4↓+H2O 按反应来看,最后产物中有NaOH,而Ba(OH)2的碱性要比NaOH的碱性要更弱,不是强碱制弱碱吗?为什么这里是这
这里遵循的是化学平衡(这个反应表面看它不是一个可逆反应,但是事实上几乎所有的化学反应都是可逆的,只是程度不一样,我们平时所说的可逆反应指的是程度较大的)的原则,也就是说,当生成物中有难溶的物质时(这个沉淀也难溶于生成的强酸或强碱),这个反应是可以发生的,这是因为化学平衡,在这个反应里,BaSO4沉淀,那么生成物中硫酸钡的在溶液中的浓度就很小了,促进了化学反应的进行.化学里面有很多这样的弱酸制强酸以及弱碱制强碱的反应.
另外,还要告诉你的是,氢氧化钡的碱性也是很强的,只不过是因为它在水中的溶解度不大,所以氢氧化钡溶液的碱性不像氢氧化钠那么强,但是事实上,氢氧化钡也是强碱.
此反应 朝离子浓度减少的方向进行
碱 + 盐另一种碱 + 另一种盐
参加反应的碱和盐必须都能溶于水, 同时生成物中必须有难溶物或容易挥发的碱(NH3•H2O)。
这里生成了BaSO4
这里并不是弱碱制强碱,你可以从离子方程式(化学反应的本质)看到其实就没有naoh生成,也就是说所谓的NAOH根本不是由BA(OH)2制出来的,只是因为ba(oh)2过量,所以一个过量的OH-与未反应的NA+拼成的,因此不叫弱碱制强碱。因为“制”的这个化学反应并没有实际发生(发没发生看离子反应)...
全部展开
这里并不是弱碱制强碱,你可以从离子方程式(化学反应的本质)看到其实就没有naoh生成,也就是说所谓的NAOH根本不是由BA(OH)2制出来的,只是因为ba(oh)2过量,所以一个过量的OH-与未反应的NA+拼成的,因此不叫弱碱制强碱。因为“制”的这个化学反应并没有实际发生(发没发生看离子反应)
收起
所谓的强碱制弱碱这说法其实是不全面的,因为咋高中化学里有很多化学式并不是以这种那么浅显的规律来进行。但是并不代表强碱制弱碱是错误的,只是它过于局限。
这个化学式中所得的Ba+、OH-、Na+都游离于溶液中,不可能说OH-和Na+结合而不和Ba+结合所以说是弱碱制强碱吧?所以说,电离方程式其实更能代表溶液中的反应实质。...
全部展开
所谓的强碱制弱碱这说法其实是不全面的,因为咋高中化学里有很多化学式并不是以这种那么浅显的规律来进行。但是并不代表强碱制弱碱是错误的,只是它过于局限。
这个化学式中所得的Ba+、OH-、Na+都游离于溶液中,不可能说OH-和Na+结合而不和Ba+结合所以说是弱碱制强碱吧?所以说,电离方程式其实更能代表溶液中的反应实质。
收起
你可以理解为溶液中一开始有全部的离子存在,但因为BaSO4是沉淀,不回到溶液中,所以反应方程式得以成立,这里不是用酸碱的电离来判断反应进行的,而是用是否生成难电解质来判断的
强电解质在水中都是以离子形式存在,在水中的反应也是离子之间的反应。反应之所以能发生,是因为生成了不溶性盐沉淀,或者电离性较低的盐或酸。
你确定氢氧化钠碱性大吗,你查查钠和钡哪个金属性强
这个问题的关键在于反应后有沉淀生成,这与强碱制弱碱不一回事,并不矛盾。
这是碱与酸式盐反应,不能用强碱制弱碱来解释!