1、___Zn+___HNO3——___Zn(NO3)2+___NH4NO3+___H2O2、___P+___HNO3+___H2O——___H3PO4+___NO↑3、————HClO3+___P+___H2O——___HCl+___H3PO44、___SO2+___KMnO4+___H2O——___K2SO4+___MnSO4+___H2SO45、___Pt+___HNO3+___HCl——___H2PtCl6+
来源:学生作业帮助网 编辑:作业帮 时间:2024/07/06 00:25:53
1、___Zn+___HNO3——___Zn(NO3)2+___NH4NO3+___H2O2、___P+___HNO3+___H2O——___H3PO4+___NO↑3、————HClO3+___P+___H2O——___HCl+___H3PO44、___SO2+___KMnO4+___H2O——___K2SO4+___MnSO4+___H2SO45、___Pt+___HNO3+___HCl——___H2PtCl6+
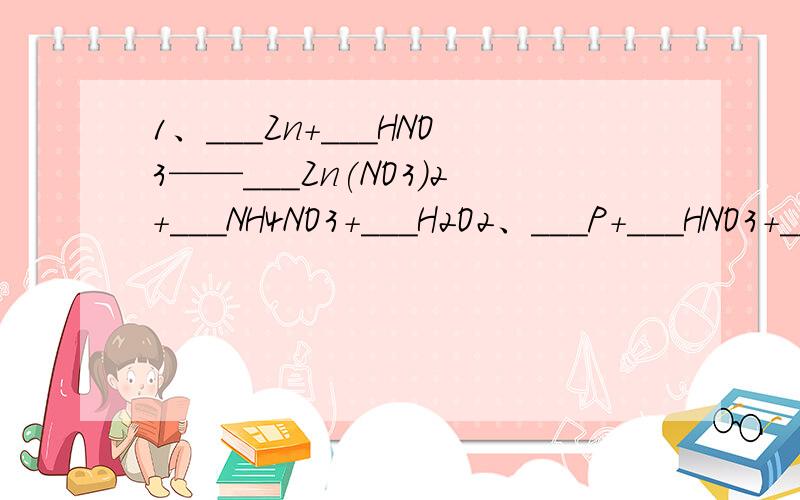
1、___Zn+___HNO3——___Zn(NO3)2+___NH4NO3+___H2O
2、___P+___HNO3+___H2O——___H3PO4+___NO↑
3、————HClO3+___P+___H2O——___HCl+___H3PO4
4、___SO2+___KMnO4+___H2O——___K2SO4+___MnSO4+___H2SO4
5、___Pt+___HNO3+___HCl——___H2PtCl6+___NO↑+___ __________
6、___Cr2O72-+___C+___H+——Cr3++___ CO2↑+___ __________
7、___Cr3++___Na2O2+___OH-——___CrO42-+___Na++___H2O
8、___Fe+___HNO3——___Fe(NO3)3+___NO2↑+___H2O
9、___Fe3O4+___HNO3——___Fe(NO3)3+___NO2↑+___H2O
10、___KI+___KIO3+___H2SO4——___K2SO4+___I2+___H2O
1、___Zn+___HNO3——___Zn(NO3)2+___NH4NO3+___H2O2、___P+___HNO3+___H2O——___H3PO4+___NO↑3、————HClO3+___P+___H2O——___HCl+___H3PO44、___SO2+___KMnO4+___H2O——___K2SO4+___MnSO4+___H2SO45、___Pt+___HNO3+___HCl——___H2PtCl6+
熟记常见元素化合价,据得失电子数相等先配平氧化剂还原剂系数,再通过观察和分析(如在酸性溶液中不能在生成物中有碳酸根/碳酸氢根氢氧根离子等)配平其它物质前的系数.
自己动手多做,别只望答案