2H2(g) + O2 (g) = 2H2O(l) △H = - 571.6 kJ/mol△H = - 571.6 kJ/mol 这里的单位是千焦每摩尔,应该就是每摩尔氢气完全燃烧放出的热量吧,为什么氢气的燃烧热又是285.8kJ/mol.
来源:学生作业帮助网 编辑:作业帮 时间:2024/07/12 13:02:15
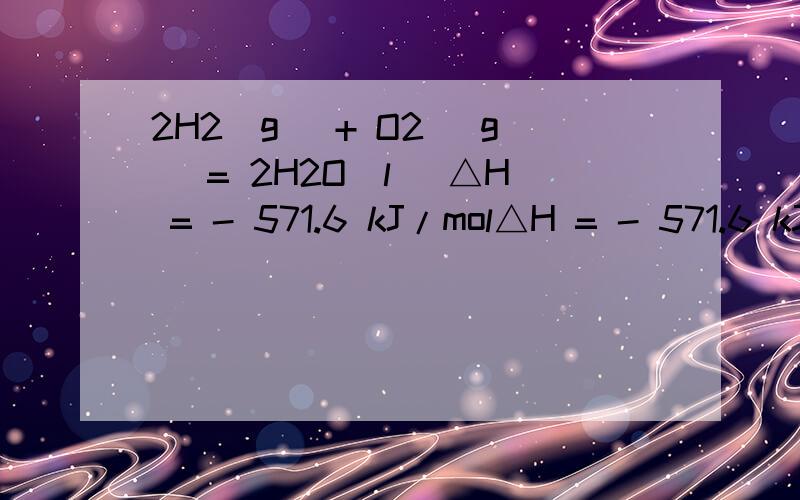
2H2(g) + O2 (g) = 2H2O(l) △H = - 571.6 kJ/mol△H = - 571.6 kJ/mol 这里的单位是千焦每摩尔,应该就是每摩尔氢气完全燃烧放出的热量吧,为什么氢气的燃烧热又是285.8kJ/mol.
2H2(g) + O2 (g) = 2H2O(l) △H = - 571.6 kJ/mol
△H = - 571.6 kJ/mol 这里的单位是千焦每摩尔,应该就是每摩尔氢气完全燃烧放出的热量吧,为什么氢气的燃烧热又是285.8kJ/mol.
2H2(g) + O2 (g) = 2H2O(l) △H = - 571.6 kJ/mol△H = - 571.6 kJ/mol 这里的单位是千焦每摩尔,应该就是每摩尔氢气完全燃烧放出的热量吧,为什么氢气的燃烧热又是285.8kJ/mol.
你用了2摩尔呀,其实你应该写成
H2(g) + 1/2O2 (g) = 2H2O(l) △H = - 285.8 kJ/mol
在101 kPa时,1 mol纯物质完全燃烧生成稳定的氧化物时所放出的热量,叫做该物质的燃烧热.单位为kJ/mol
燃烧热是以1 mol可燃物作为标准来进行测定的,因此在计算燃烧热时,热化学方程式里其他物质的化学计量数常出现分数:如H2(g)+ 1/2O2(g)====H2O(l);ΔH=-285.8 kJ·mol-1 这时的分数是代表摩尔数而不是分子个数所以是合理的.注意 化学方程式中ΔH为负值 而在叙述时 用正值描述
△H = - 571.6 kJ/mol 单位是千焦每摩尔,请注意反应式中H2的系数是2不是1。这表示2mol氢气完全燃烧的放热量!!所以氢气的燃烧热是285.8kJ/mol。
LZ很对啊,燃烧热是每摩尔的物质完全燃烧放出的热,那个方程里氢气是2mol啊
燃烧热的定义是1mol物质完全燃烧,上述式子是哪个反应的反应热